(1)9.9×25,=(10-0.1)×25,=10×25-0.1×25,=250-2.5,
来源:学生作业帮 编辑:大师作文网作业帮 分类:数学作业 时间:2024/10/03 04:31:34
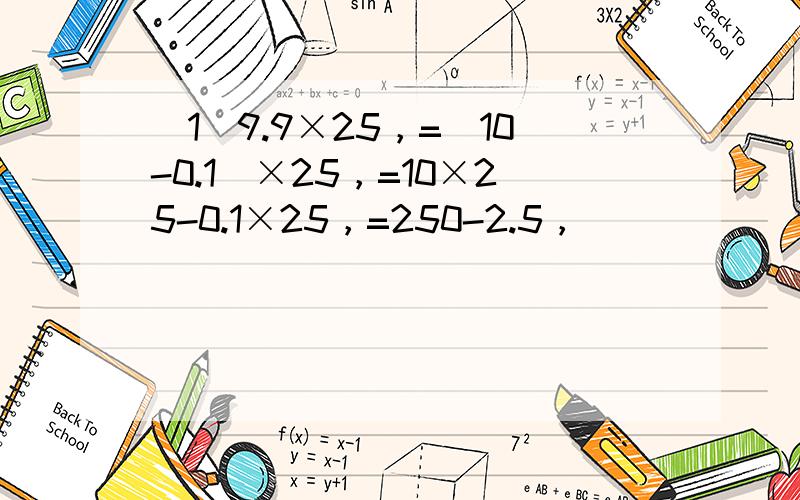 (1)9.9×25,
=(10-0.1)×25, =10×25-0.1×25, =250-2.5, =247.5; (2)0.75×0.5×4×0.2, =(0.75×4)×(0.5×0.2), =3×0.1, =0.3; (3)1.25×32×0.25, =1.25×8×4×0.25, =(1.25×8)×(0.25×4), =10×1, =10; (4)8.12×4+1.88×4, =(8.12+1.88)×4, =10×4, =40; (5)0.32×13.4-0.32×3.4, =0.32×(13.4-3.4), =0.32×10, =3.2; (6)3.84×99+3.84, =3.84×(99+1), =3.84×100, =384; (7)2.009×390+20.09×41+200.9×2, =20.09×39+20.09×41+20.09×20, =20.9×(39+41+20), =20.9×100, =2090; (8)(9.57+9.57+9.57+9.57 )×25, =(9.57×4)×25, =9.57×(4×25), =9.57×100, =957.
H2A=H++HA-,HA-=H++A2-(25度Ka=10-2),在0.1mol/l的H2A溶液中C(H+)大于0.1
由0.1mol.l一元碱BOH溶液的PH=10可知为弱碱为什么(25度)
由0.1mol.l一元碱BOH溶液的PH=10克制为弱碱为什么(25度)
(-0.125)^25×(-8)^25=( ) 0.75^10×(-4/3)^10=( )
[10+(1-5/1-65%)/1]*0.4 (4/3=6/1=8/3)*2.4+9.9 525*48/(3*8*25)
25℃时,0.1mol/L氨水溶液中c(H+)c(OH−)=1×10-8,下列叙述正确的是( )
H2A=H++HA-,HA-=H++A2-(25度Ka=10-2),问为什么在0.1mol/L的Na2A的溶液中,C(A
25/()=0.625
(0.125)^25×(-8)6^25=? 0.75^10×(-4/3)^10?
25乘9又8分之7 =25乘10减25乘() =()
10÷(22+X)=25(解方程)
(1)25℃时,0.1mol•L-1的HA溶液中c(OH-)=10-10 mol/L,0.01mol•L-1的BOH溶液
|