The specific heat capacity of silver is 0.24 J/°C·g.
来源:学生作业帮 编辑:大师作文网作业帮 分类:英语作业 时间:2024/09/21 13:32:44
The specific heat capacity of silver is 0.24 J/°C·g.
(a) Calculate the energy required to raise the temperature of 180.0 g Ag from 273 K to 303 K.
_____________J
(b) Calculate the energy required to raise the temperature of 1.0 mol Ag by 1.0°C (called the molar heat capacity of silver).
_____________J/mol°C
(c) It takes 1.05 kJ of energy to heat a sample of pure silver from 12.0°C to 14.7°C.Calculate the mass of the sample of silver.
_____________g
(a) Calculate the energy required to raise the temperature of 180.0 g Ag from 273 K to 303 K.
_____________J
(b) Calculate the energy required to raise the temperature of 1.0 mol Ag by 1.0°C (called the molar heat capacity of silver).
_____________J/mol°C
(c) It takes 1.05 kJ of energy to heat a sample of pure silver from 12.0°C to 14.7°C.Calculate the mass of the sample of silver.
_____________g
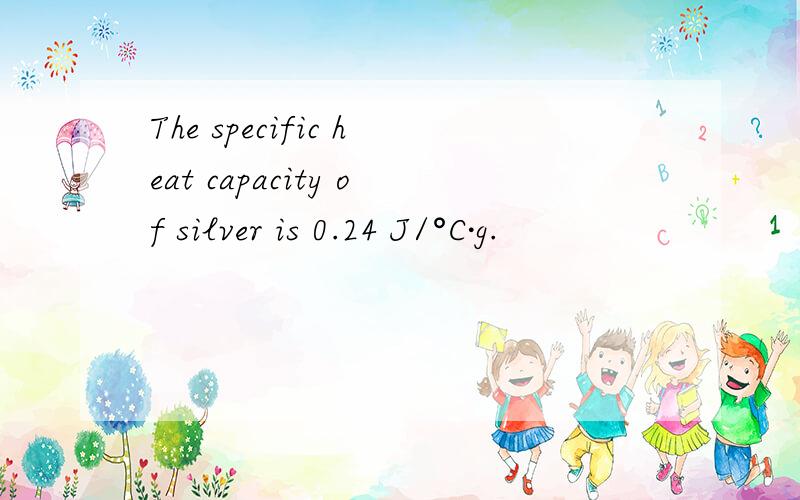
(a)180.0*(303-273)*0.24=1296J
(b)1.0 mol Ag是108g,所以 108*1*0.24=25.92J
(c)1.05*1000/(14.7-12.0)再/0.24=162.04g
(b)1.0 mol Ag是108g,所以 108*1*0.24=25.92J
(c)1.05*1000/(14.7-12.0)再/0.24=162.04g
The specific heat capacity of silver is 0.24 J/°C·g.
the heat capacity of water is relatively high.这里的is relative
what is the capacity of carbon dioxide?
翻译四句话.The oceans have a very big heat capacity because of th
Which one has Higher specific heat capacity Ice ,water or ga
请问一道英文化学题Calculate the specific heat of a substance given th
英语翻译the dissolution capacity of the solvent is directly link
什么是水的热容量“heat capacity of water”
molar heat capacity是什么意思
Silver is the best conductor of electricity,copper it closel
Silver is the best conductor of electricity, copper________
The Lining Is Silver 歌词